Solving Human Health Challenges Through Preclinical Expertise & Innovation
Who We Are
We exist to advance human health, and are committed to helping advance solutions to complex diseases. From our commitment to knowing and improving the human condition comes our name: Virscio (to know “scio” humans “vir”).
As a specialized AAALAC accredited, GLP-compliant preclinical CRO, Virscio engages with leading academic institutions, venture and private equity-backed biotechnology companies, and global pharmaceutical companies to help design and execute capital efficient science that is focused on de-risking elements of drug development that often contribute to failed clinical trials.
Pursuing a science-first focus, we leverage 30+ years of preclinical experience, along with deep datasets in our nonhuman primate test system, to inform thoughtful, value-add preclinical study designs that yield definitive, actionable data to inform program decision making.
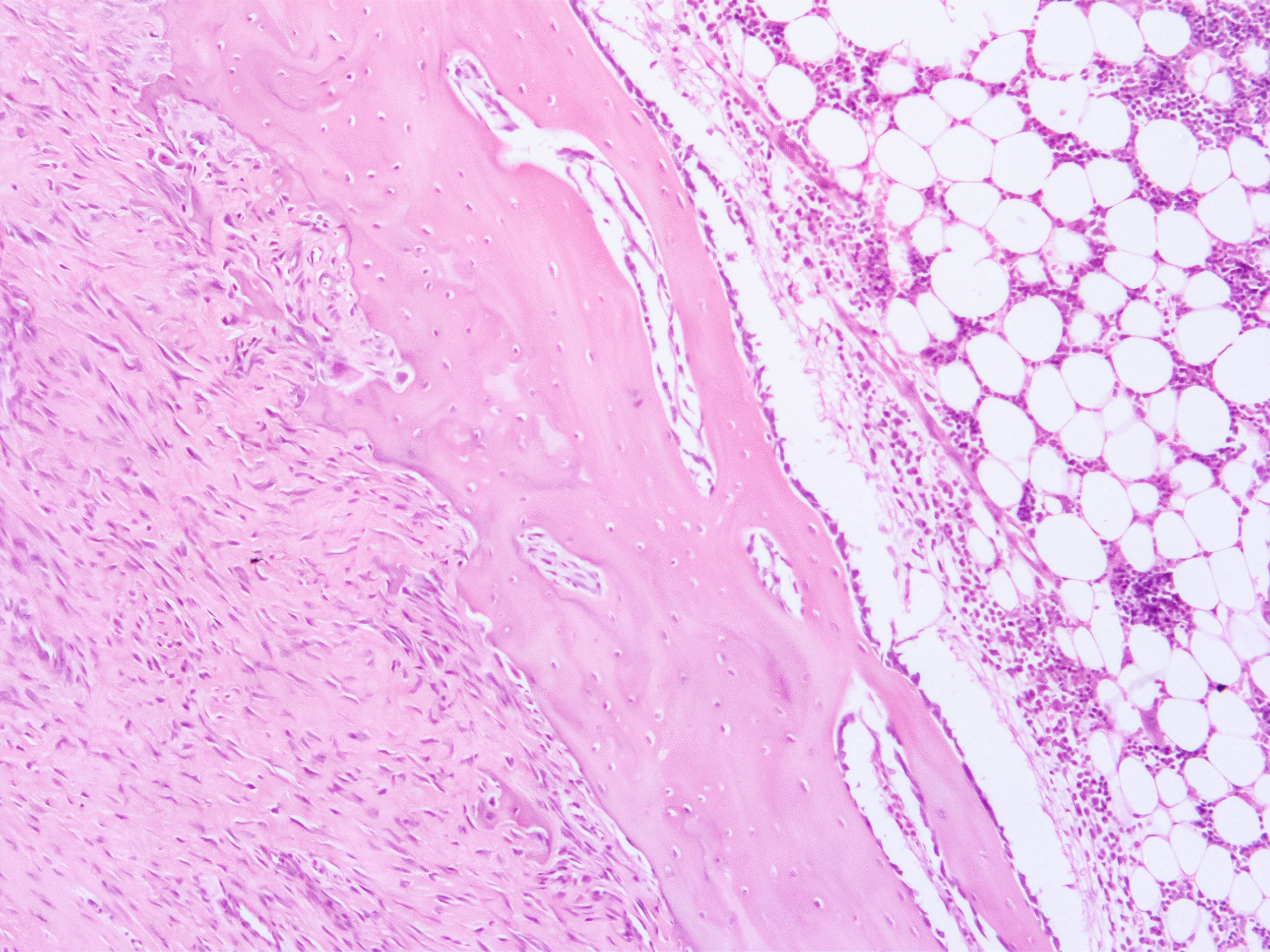
What We Do
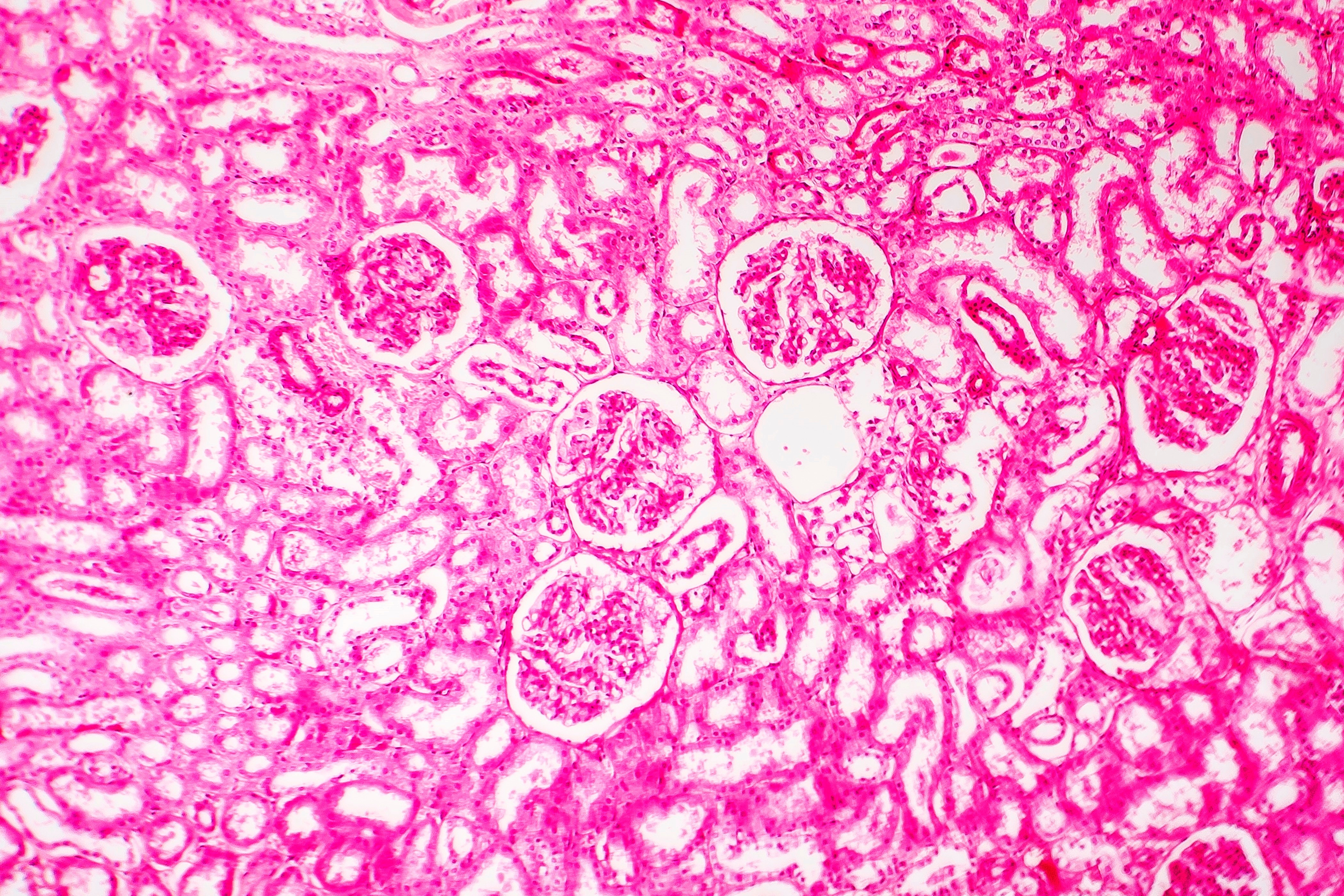
We address translational risks by applying best understanding of human pathophysiology to inform the execution of non-GLP and GLP studies designed to evaluate delivery, pharmacokinetics, pharmacodynamic, safety, and efficacy of candidate therapeutics.
As stewards of a globally unique nonhuman primate resource, we maintain the very highest ethical and scientific standards to drive the best possible study designs, data analysis, and animal care and use. We embrace these responsibilities and are recognized internationally for this commitment.

Differentiating Expertise, Capabilities, & Resources
Virscio represents a multi-decade strategy to enable more efficient innovation, while elevating animal welfare and lowering the overall cost to successfully take a therapeutic program from concept to clinic.
- Fully integrated in vivo, histopathology, and molecular research cores, enabling seamless engagement and service delivery
- MD and PhD Subject Matter Experts (SMEs) leading engagement, scientific strategy and protocol development to ensure study execution is aligned to development objectives
- Deep scientific and clinical expertise in ophthalmology, CNS, cardiovascular, and metabolism
- Extensive portfolio of validated histomolecular assays to support both primate and non-primate
- Dedicated IACUC, enabling thorough time efficient protocol review and approvals
- Expedited initiation timelines, with flexibility to accommodate schedule changes
- Skilled Study Directors with extensive NHP study execution experience
- Flexible, and efficient protocol and contract amendment process
- Robust data analysis and study reporting, and commitment to generating definitive, actionable study outcomes and conclusions
- Humane, sustainable access to a large, biologically clean nonhuman primate population

Testimonials
Our Leadership
Matthew Lawrence, M.D., Ph.D.
Chief Executive Officer, Chief Scientific Officer

Christopher Stanley
Chief Business Officer, Chief Financial Officer

Our Affiliations
In addition to serving the R&D needs of innovative organizations around the world, Virscio is a proud member of many industry organizations that share our mission of advancing biomedical innovation and improving clinical outcomes.
- AAALAC
- NIH
- Alliance for Regenerative Medicine
- National Association for Biomedical Research
- Biotechnology Industry Organization (BIO)
- Biocom
- CIRM
- ARPA-H