Research Services
Virscio is a leading provider of nonhuman primate data and histomolecular services to effectively guide your programs and candidates to clinical success.
From preclinical proof of concept to IND-enabling GLP safety studies, Virscio’s goal is to bring clarity and confidence to R&D investment decision making and improve the odds of clinical translation.
We fully support critical development stages, spanning preclinical strategy, protocol design, in-life study execution, sample processing, data analysis, study reporting and defining regulatory path, with a focus on in vivo primate efficacy and safety, pharmacokinetics, pharmacodynamics and therapeutic delivery.
Virscio additionally performs advanced histopathology and molecular analysis in support of both primate and non-primate preclinical studies. Our capabilities are enabled by extensive experience.
Translational Experience
... and globally distributed pharmaceutical, biotechnology and academic sponsors
Efficacy Models
Virscio has established nonhuman primate models that emulate human disease pathology to evaluate efficacy of small molecules, biologics, gene and cell therapies, medical devices and delivery technologies.
Applied models meet these critical induction, characterization and validation criteria:
- The clinically relevant disease pathology can be reproducibly induced within variability limits that allow candidate screening protocols of sufficient statistical power to permit discovery and development decisions.
- The induced disease pathology can be modulated by a clinical standard of care intervention and/or the therapeutic target is present in the induced disease phenotype and can be measured or inferred through validated, correlated endpoints.
- The disease pathophysiology can be induced and sustained with limited animal welfare impact.
Ophthalmology
- Wet age-related macular degeneration
- Retinal epithelium and photoreceptor degeneration
- Retinal neovascularization
- Glaucoma
- Uveitis
- Optic neuropathy
- Surgical inflammation
- Myopia
- Amblyopia
- Cataract
Central Nervous System
- Alzheimer’s disease
- Parkinson’s disease
- Stroke
- Spinal cord injury
- Schizophrenia
- Dystonia
- Behavior and cognition
Cardiovascular
- Hypertension
- Heart failure
Metabolism
- Diet-induced obesity
- Hyperlipidemia
Immunology
- Vaccine and adjuvant development
- Immunomodulation
Delivery
- MRI and ultrasound guided
- Sustained release implants and devices
- Gene therapy vectors
- Cell therapies
- All routes for small molecules and biologics
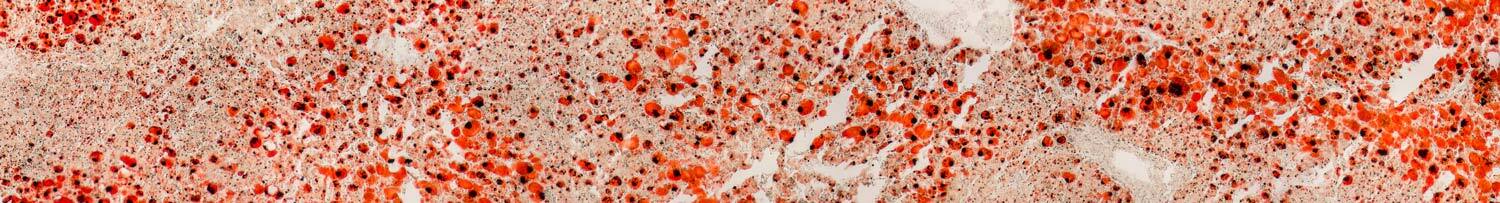
Our Research Solutions
Virscio’s unique, unparalleled access to a biologically clean primate population allows timely initiation of studies in treatment naïve or non-naïve animals of preferred age range, sex, weight and number, guided by specific research objectives.
By employing clinically relevant primate models, Virscio drives accurate characterization of human risk and response to therapeutic candidates to speed candidate selection and improve clinical success. Subject selection, model optimization and well characterized quantitative endpoints further allow reduction and refinement of animal use.
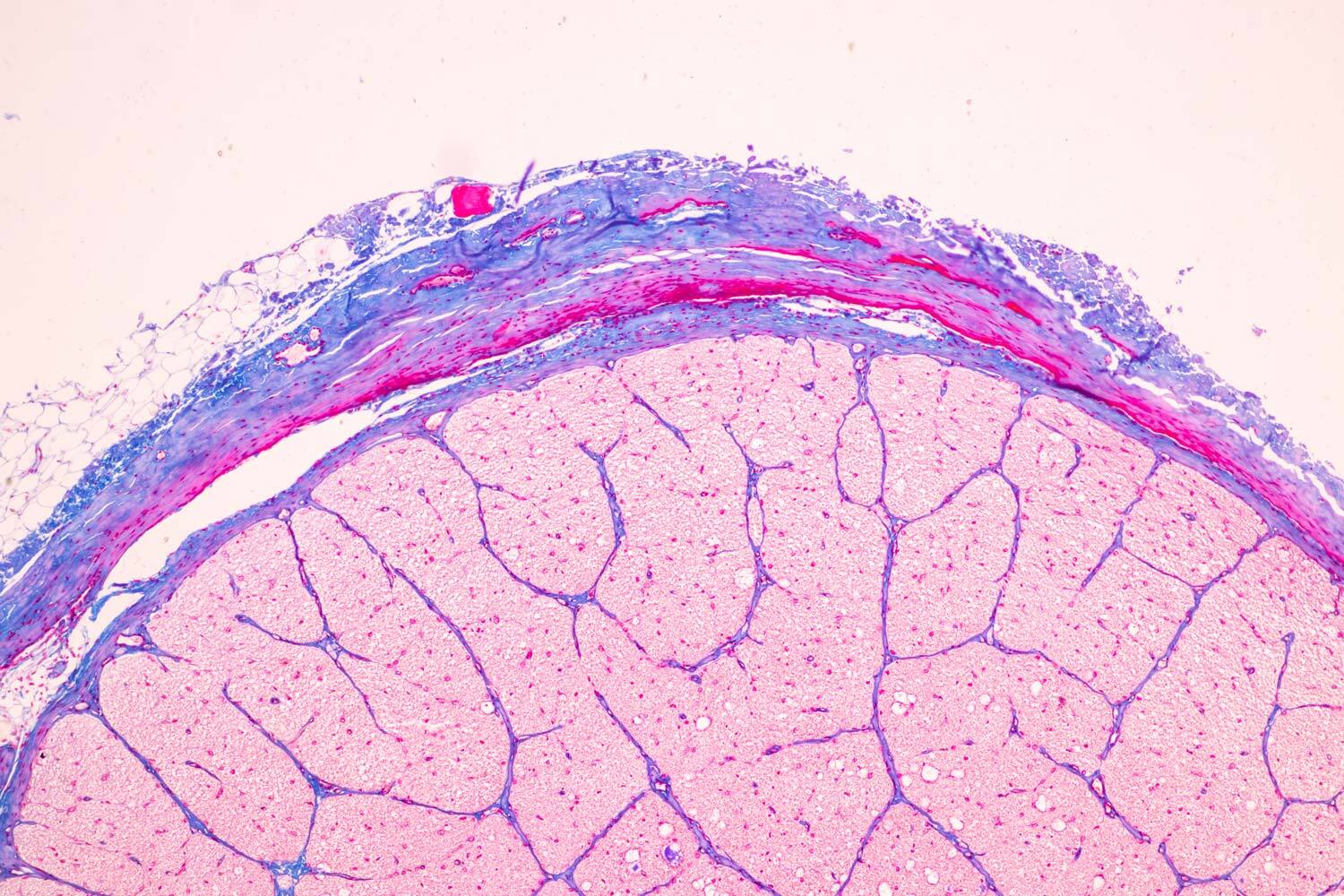
Study Designs Conducted
- Pharmacokinetics (PK)
- Pharmacodynamics (PD)
- Delivery Optimization
- Safety Pharmacology
- GLP Toxicology (acute and chronic)
Routes of Administration
Systemic
-
Oral
-
Subcutaneous
-
Intramuscular
-
Intravenous
-
Transdermal
-
Nasal
CNS
-
Intraventricular
-
Intrathecal
-
Intraparenchymal
-
Subpial
Ophthalmic
-
Topical ophthalmic
-
Punctal
-
Subconjunctival
-
Intracameral
-
Intravitreal
-
Subretinal
-
Suprachoroidal
Gene and Cell Therapy Evaluations
- Neutralizing antibody guided recruitment
- Target tissue delivery
- Detailed biodistribution determination
- Cell survival assessment
- Immunosuppression regimens
- Pharmacodynamic endpoints
- Functional endpoints
- GLP safety
Sampling
- Serum
- Plasma
- Whole blood
- Peripheral blood mononuclear cells
- Cerebrospinal fluid
- Urine
- Aqueous humor
- Vitreous humor
- Tissue biopsies
- Tissue dissociation for single cell RNA-Seq
- Comprehensive tissue collection
Surgical Modeling
- Ophthalmic
- Neurosurgical
- Cardiac
- Renal
- Orthopedic
- Stereotaxic
- MRI-guided
- Telemetry
- Device Implantation
Safety Endpoints
- Ocular
- Central nervous system
- Systemic
- Clinical pathology
- Histopathology
- Imaging
- Electrophysiology
- Functional observational batteries
- Toxicokinetics
- Therapy-specific endpoints
Scheduling and Excecution
- Accelerated initiation
- Phased study designs
- Single, repeat and chronic dosing
- Onsite quality assurance
Discover more about our capabilities
Visit our scientific library and get access to a wide range of resources showcasing Virscio’s translational research capabilities, including posters, case studies, journal articles and more.
Histopathology
Virscio’s histopathology and histomolecular experience fully supports both nonhuman primate studies and studies conducting in other non-primate species.
Quantitative assessment of critical safety and tissue pharmacodynamic endpoints are achieved through core established capabilities, robust assay validation, image analysis, and integration of orthogonal data.
We apply our expertise to help guide your histopathology strategy and objectives.
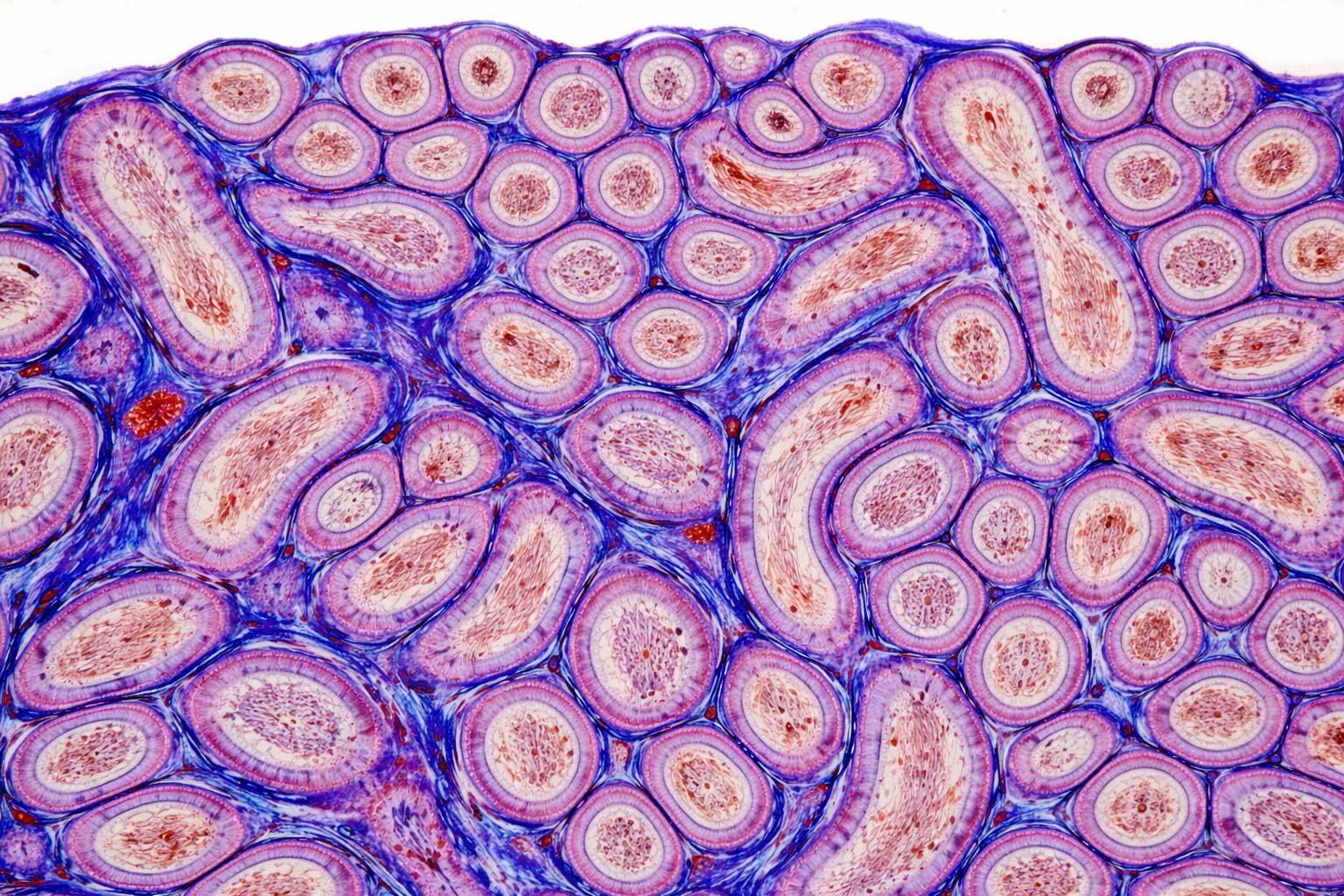
Discovery Histopathology
- Target distribution and indication selection
- Tissue distribution of AAVs, lipid nanoparticles, other viral and non-viral vectors, stem cells, therapeutic antibodies, tag proteins (GFP, RFPs, FLAG, V5, HA)
- Rigorous immunohistochemistry assays development using 20+ tissue matrices correlated with single cell RNA-Seq and public omic databases
- Model characterization, validation and efficacy assessment referencing established historical control databases
Toxicologic Histopathology
- Non-GLP and GLP nonclinical safety primary interpretation and peer-reviews in African green monkey, cynomolgus monkey, mouse, rat, dog, pig, human, and other test systems
- Mechanistic toxicology, including companion molecular methods (RNASeq, western blot, qRT-PCR, ELISA)
- Briefing books and IND authoring, representations at type C meetings, and responses to FDA comments
Histopathology Tools
- Specialty and customized trimming of complex tissues (e.g., eye, kidney, heart, brain)
- Processing to paraffin and frozen sections
- Extended catalog of special stains
- Extended catalog of optimized immunohistochemistry assays
- Whole mount immunohistochemistry (IHC)
- In situ hybridization
- Multiplex IHC
- Duplex IHC/ISH
- Duplex ISH
- Visiopharm image analysis with AI app development
- Secured digital histology platform (Concentriq for Research)
- Data integration of clinical pathology, circulating and tissue biomarkers, omics, and in vivo readouts
- Custom tissue microarrays assembly including cell pellets and tissues
- Western blots, ELISA, qRT-PCR
- Nonhuman primate tissues of diverse age with and without test article exposure
- In-house quality assurance unit (QAU) supports GLP studies accommodating .svs, .mrxs, .ndpi, and other Visiopharm supported file formats
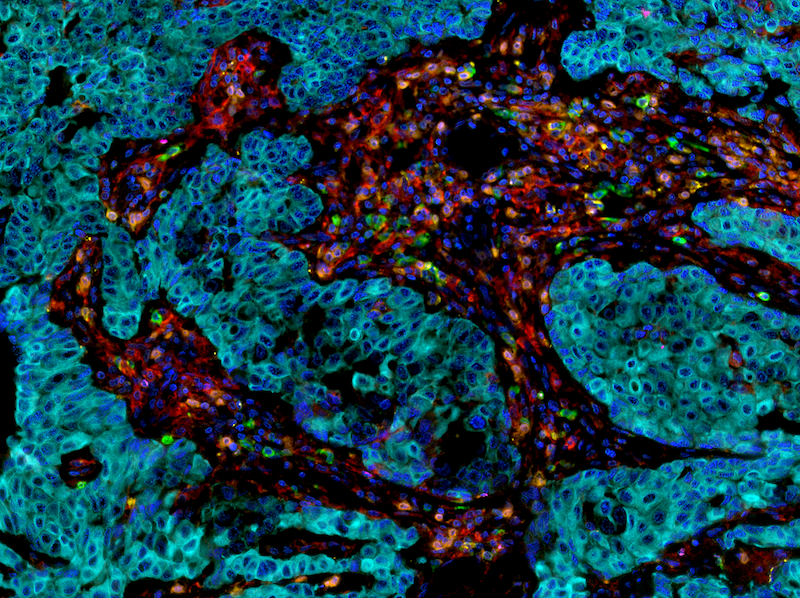
Multiplex IHC
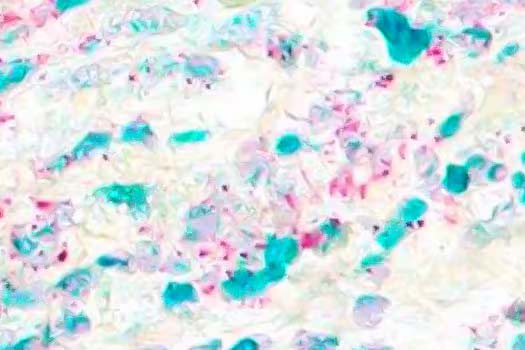
Duplex IHC/ISH
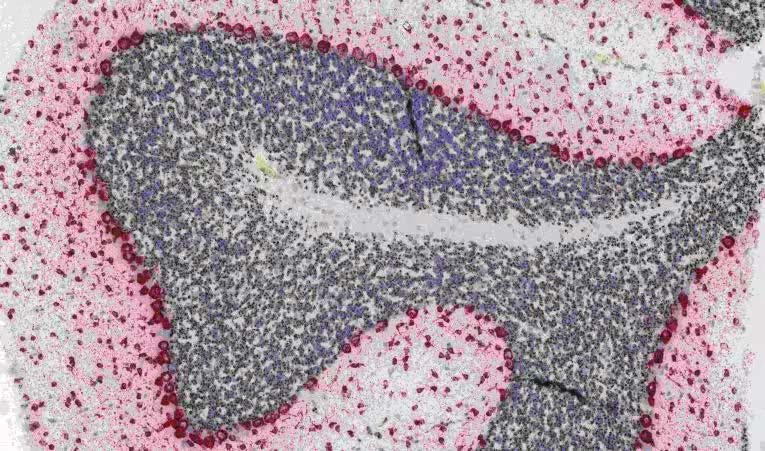
Duplex ISH
Therapeutic Areas
- Ophthalmology
- Neurology
- Cardiovascular
- Immunology/inflammation
- Metabolic
- Oncology
- Musculoskeletal
- Renal/urinary
- Gastrointestinal
- Skin
- Respiratory
Molecular Biology
We work with you to develop molecular analysis strategies to optimize scientific and budgetary scope and objectives.
Custom method development and validation
- Bespoke method and test system development and validation to achieve data objectives
- Analysis of all tissue types, including liver, spleen, kidney, brain, eye, and difficult to process tissues
High-throughput tissue sample processing
- DNA, RNA and protein isolation from tissue, blood, biofluids and cultured cells
- Isolation and quantification using Agilent 2100, Spectra Max, Qubit, and Nanodrop
Biodistribution assays
- Custom biodistribution assay development for multiple tissues, including heart, liver, brain, and eye
- Sample analysis using qPCR and ddPCR for WPRE, ITR2, GAPDH, TERT, GFP, Beta-ACTIN
ELISA assays
-
Validation of custom or commercially available ELISA assays and sample analysis
Primary cell isolation
- Isolation of PBMCs, hepatocytes, fibroblasts and other cell types


Gene expression profiling
- Comprehensive DNA and RNA quantification, quality assessment
and stability analysis - Expression profiling for study specific genes and genes of common interest (LIF, LEPTIN, CNTF, IL6, GFAP, C3, VIMENTIN, RLBP1, FGF2, EDN2, TNF)
- Absolute and relative quantification through qPCR and ddPCR
Western blots
- Method development, assay validation, and high throughput analysis for study specific proteins and proteins of common interest (MAPT, NEFL, GFAP, VIMENTIN, TUBULIN, C3, and Beta-ACTIN)
Tissue dissociation and library preparation for single cell sequencing
- Single cell dissociation and library preparation for PBMC, CNS, retinal and other cell types for Parse and 10x Genomics sequencing protocols
Data capture and reporting
- Robust program management and electronic data capture to meet documentation, regulatory, and timeline needs
VIRSCIO RESEARCH SERVICES
