Resources
Facilities & Infrastructure
Virscio maintains state-of-the-art research facilities at two locations to meet client needs.
New Haven, CT
Virscio’s US headquarters is the location of histopathology operations, sample banking, logistics, finance, IT, and administrative functions supporting in vivo, histopathology, and molecular divisions.
St. Kitts, West Indies
Virscio’s nonhuman primate vivarium, in-life study operations, and molecular analyses are based in St. Kitts, West Indies, a highly accessible and hospitable primate source country, home to an abundance of introduced African green monkeys.

Research Operations
- GLP-compliant since 2008, AAALAC-accredited since 2010
- 125+ trained Study Directors, veterinarians, technicians, and support staff
- Recognized by commercial, NIH, and academic sponsors for world-class platform
- Consistently commended by AAALAC for exceptional animal welfare program

- Independent, dedicated, and responsive IACUC committee
- Extensive sample management, handling, and global shipping capabilities
- Government conservation partner, providing humane monkey population control
- Committed to staff training, education, and career development
Infrastructure
- 2,500+ annual animal capacity, with ongoing expansion
- Dedicated animal quarantine, social housing, and testing infrastructure
- Surgery rooms, laboratories, and imaging suites
- Extensive in vivo imaging capabilities
- Powered by renewable energy, with water and electrical redundancies

Imaging
- Ultrasonography
- Echocardiography
- MRI/MRA
- DEXA
- X-ray
- cSLO/OCT/OCTA
- Fundus imaging
- Angiography
- Specular microscopy
- Slit lamp biomicroscopy
- Indirect ophthalmoscopy
- Gonioscopy
- Endoscopy
- Intraoperative video
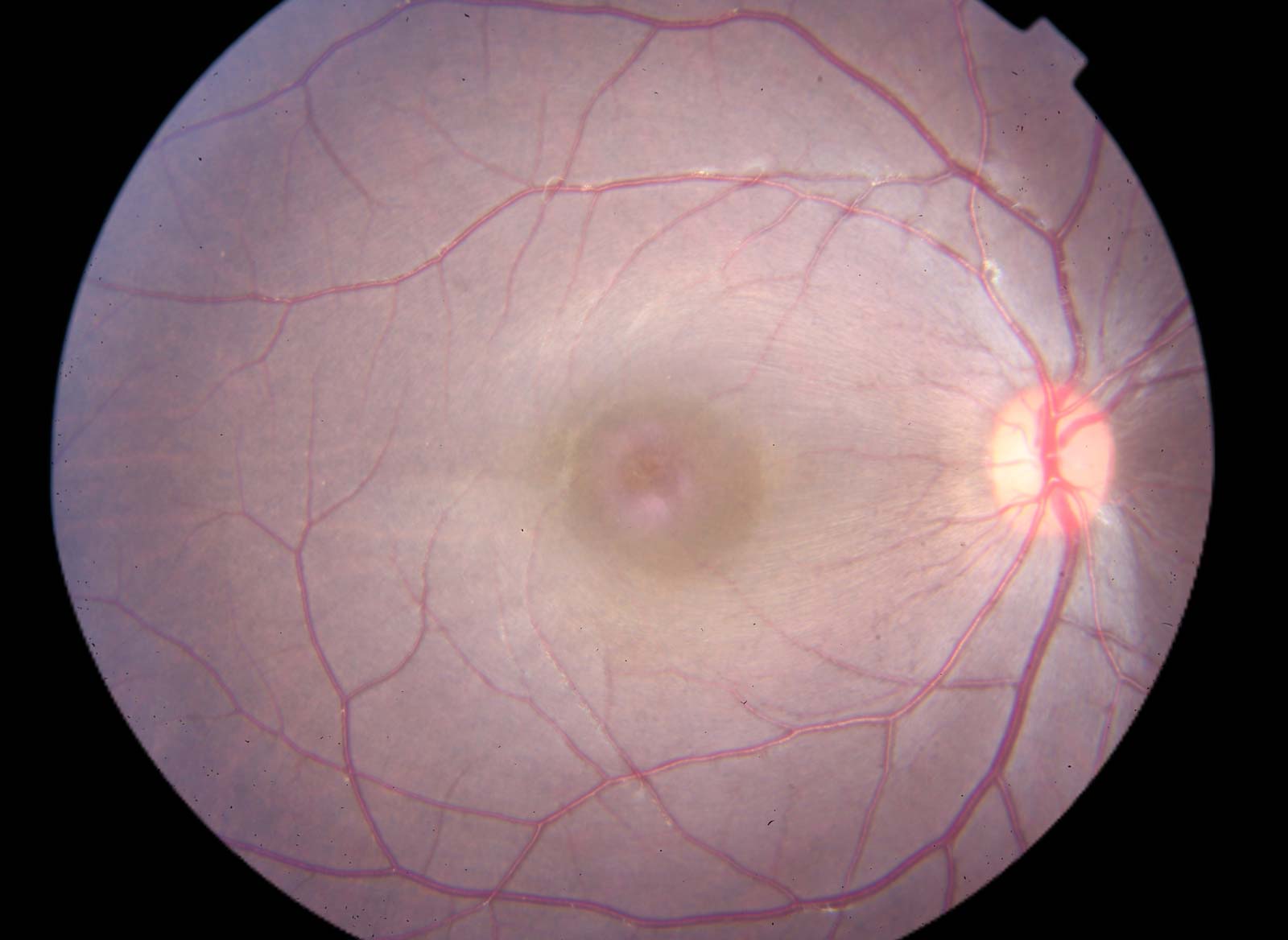
Ophthalmic Imaging
- Fundus photography
- Flourescein angiography
- Optical coherence tomography
- Specular microscopy
- Slit lamp ophthalmoscopy
- Direct ophthalmoscopy
- Gonioscopy
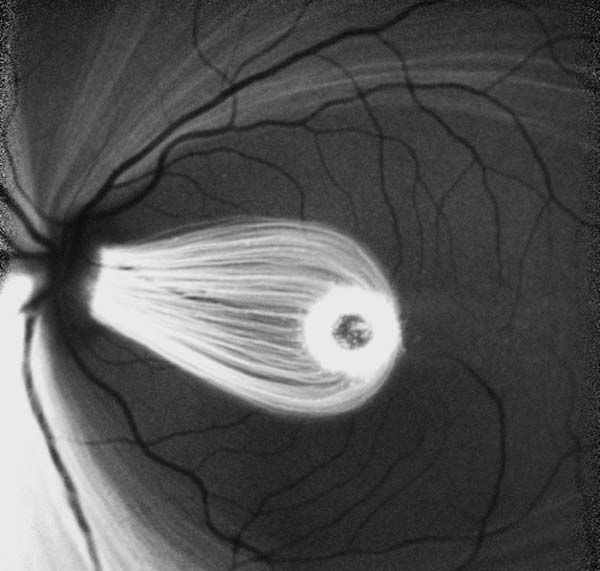
Ophthalmic Physiology and Model Induction
- A scan ultrasound
- Fluorophotometry
- Laser flare photometry
- Full-field electrophysiology
- Multifocal electrophysiology
- Pachymetry
- Tonometry
- Keratometry
- Autorefraction
- 532nm laser
- Phacoemulsification
- Vitrectomy
- Surgical microscopy
- Endoillumination
- Micro-instrumentation
Telemetry, Kinematics, and Cognition
- Physiological telemetry
- Cardiac telemetry
- EEG telemetry
- Pedography
- Dynamometry
- Activity monitoring
- Gait kinematics
- WGTA cognitive testing
- CANTAB cognitive testing
- Functional observation batteries (FOBs)
- Jacketed external telemetry (JET)
Surgery and Specimen Collection and Handling
- Stealth S7 Surgical Navigation
- Microsurgical instrumentation
- Stereotaxic instrumentation
- Cell culture
- Biosafety containment
- Cryopreservation
- Fine tissue dissection
- Liquid nitrogen cold chain
- Global shipping logistical support
Clinical Pathology and Biomarker Assays
- Clinical chemistry
- Complete blood counts
- Coagulation profile
- ELISA
- Western blot
- qPCR/RT-qPCR
- Spectrophotometry
- Flow cytometry
- DNA methylation
- Histology
- Immunohistochemistry
- In situ hybridization
Physiology and Anatomic Measures
- Body weight
- Body temperature
- Respiratory rate
- Blood pressure
- Heart rate
- Electrocardiograms
- Tonometry
- Pachymetry
- Flare photometry
- Keratometry
- A-scans
- Electroretinography
- Full and multifocal ERG
- Visual evoked potentials
Discover more about our capabilities
Visit our scientific library and get access to a wide range of resources showcasing Virscio’s translational research capabilities, including posters, case studies, journal articles and more.
Biological Resources
African green monkeys (also called vervets) are a non-endangered, Old World nonhuman primate (NHP) species. A subpopulation of green monkeys was introduced to St. Kitts, West Indies in the 1600s where they now thrive and present an agricultural threat as an invasive species without natural predators.

Green monkeys are genetically similar to cynomolgus and rhesus macaques and evolutionarily equidistant to humans, with significant shared physiology, anatomy and related comparative biology. Derived from a small founding population, the degree of homozygosity of St. Kitts green monkeys accommodates study designs of smaller sample size. Investigative new drug and device filings supported by green monkey data are accepted by the FDA, EMA and other regulatory agencies for clinical trial approvals.
With the absence of zoonoses common to continental NHP populations and a genetically constricted founding population, the St. Kitts green monkey represents a unique, biologically clean NHP resource for in vivo and ex vivo research at academic, government and industry laboratories internationally, and the conduct of translational and therapeutic development studies by Virscio scientists in St. Kitts with industry and academic sponsors and collaborators.
St. Kitts green monkeys have additionally been a source of NHP cell lines for vaccine development and in vitro assays. Green monkey derived Cos-7 and Vero cells are widely used as a well-characterized platform for in vitro analyses.
Comparative NHP Biology
Nonhuman primates (NHPs) provide a critical translational bridge in the extension of in vitro and rodent findings to the understanding of human physiology and disease.
As an Old World primate the green monkey is among the most genetically and physiologically homologous research species to humans and most studied.
The translational value of the St. Kitts green monkey is supported by a reference genome, and comprehensive neuroanatomic resources.
These databases, along with existing and ongoing genotyping and phenotyping of the species in healthy and disease states at Virscio and other institutions has established the St. Kitts green monkey as among the most comprehensively characterized NHP for systems biology and genomic and epigenomic research to inform understanding of human disease and therapeutic response.
 Historical biomedical applications of NHP test systems (and other animal model systems) have been driven by availability of animals and background control data.
Historical biomedical applications of NHP test systems (and other animal model systems) have been driven by availability of animals and background control data.
Indian-derived rhesus macaques, which were readily accessible at the time of early polio vaccine development were used extensively for this urgent need and adopted for broader biomedical purposes. Rhesus monkeys continue to be a core resource for U.S. national primate centers. However, tightened access to rhesus macaques with geopolitical shifts directed attention to cynomolgus macaques and African green monkeys.
Over time cynomolgus monkeys were more heavily adopted for ease of access to wild Southeast Asian populations and the perceived higher risk of zoonosis in African-derived green monkeys. While this contributed to extensive background control data development for cynomolgus monkeys, particularly for safety endpoints, comparative clinical relevance over other primate species was not a principal driver to selection of cynomolgus monkeys as a test system.
With broadening application of NHPs to therapeutic development, normative datasets have emerged for African green monkeys, establishing the substantial comparability to macaque species across many parameters. Homology analyses of the green monkey reference genome, derived from St. Kitts-origin animals, further confirms the green monkey to be evolutionarily equidistant to humans, with loci of tighter genome and proteome homology than macaques.
Unlock the Potential of African Green Monkeys in Your Research
Dive into the world of preclinical research with exclusive access to our comprehensive African Green Monkey datasets. Virscio invites you to explore how this unique nonhuman primate species can be pivotal in advancing your research endeavors.
Publications
LNA-mediated microRNA silencing in non-human primates
Nature, 2008
→ View Publication
Pharmacological inhibition of a microRNA family in nonhuman primates by a seed-targeting 8-mer antimiR
Science Translational Medicine, 2013
→ View Publication
Preclinical evaluation of ADVM-022, a novel gene therapy approach to treating wet age-related macular degeneration
Molecular Therapy, 2019
→ View Publication
Biodegradable scaffolds promote tissue remodeling and functional improvement in non-human primates with acute spinal cord injury
Biomaterials, 2017
→ View Publication
Optimization of laser-induced choroidal neovascularization in African green monkeys
Experimental Eye Research, 2011
→ View Publication
Exploration of the African green monkey as a preclinical pharmacokinetic model: Oral pharmacokinetic parameters and drug-drug interactions
Xenobiotica, 2009
→ View Publication
Additional Publications
- Ward K, Coon D,, Magiera D, Bhadresa S, Nisbett E, Lawrence M, Exploration of the African green monkey as a preclinical pharmacokinetic model: intravenous pharmacokinetic parameters, Drug Metabolism and Disposition, 36:715-20, 2008.
- Pritchard C, Slotkin J, Yu D, Dai H, Lawrence M, Bronson R, Reynolds F, Teng Y, Woodard E, Langer R. Establishing a model spinal cord injury in the African green monkey for the preclinical evaluation of biodegradable polymer scaffolds seeded with human neural stem cells, Journal of Neuroscience Methods, 188:258-69, 2010.
- Liddie S, Goody R, Valles R, Lawrence M. Clinical chemistry and hematology values in a Caribbean population of African green monkeys. Journal of Medical Primatology, 39:389-98. 2010.
- Glogowski S, Ward K, Lawrence M, Goody R, Proksch J. The use of the African green monkey as a preclinical model for ocular pharmacokinetic studies. Journal of Ocular Pharmacology and Therapeutics, 28:290-8, 2012.
- Cloutier F, Lawrence M, Goody R, Lamoureux S, Al-Mahmood S, Colin S, Ferry A, Conduzorgues J, Hadri A, Cursiefen C, Udaondo P, Viaud E, Thorin E, Chemtob S. Antiangiogenic activity of aganirsen in nonhuman primate and rodent models of retinal neovascular disease after topical administration, Investigative Ophthalmology & Visual Science, 53:1195-203, 2012.
- Rottiers V, Obad S, Petri A, McGarrah R, Lindholm M, Black J, Sinha S, Goody R, Lawrence L, deLemos A, Hansen H, Whittaker S, Henry S, Brookes R, Najafi-Shoushtari S, Chung R, Whetstine J, Gerszten R, Kauppinen K, Näär A. Pharmacological inhibition of a microRNA family in nonhuman primates by a seed-targeting 8-mer antimiR, Science Translational Medicine, 5(212):212ra162 2013.
- Halley P, Kadakkuzha B, Ali Faghihi M, Magistri M, Zeier Z, Khorkova O, Coito C, Hsiao J, Lawrence M, Wahlestedt C. Regulation of the apolipoprotein gene cluster by a long noncoding RNA, Cell Reports, 6:222-230 2013.
- Sidman R, Li J, Lawrence M, Hu W, Musso G, Giordano R, Cardó-Vila M, Pasqualin Ri, Arap W. The peptidomimetic Vasotide targets two retinal VEGF receptors and reduces pathological angiogenesis in murine and nonhuman primate models of retinal disease. Science Translational Medicine, 7:309ra165. 2015.
- Hsiao J, Yuan T, Tsai M, Lu C, Lin Y, Le M, Lin S, Chang F, Liu Pimentel H, Olive C, Coito C, Shen G, Young M, Thorne T, Lawrence M, Magistri M, Faghihi M, Khorkova O, Wahlestedt C. Upregulation of haploinsufficient gene expression in the brain by targeting a long non-coding RNA Improves seizure phenotype in a model of dravet syndrome. EBioMedicine. 9:257-77 2016.
- Liddie S, Okamoto H, Gromada J, Lawrence M. Characterization of glucose-stimulated insulin release protocols in African green monkeys (Chlorocebus aethiops). J Med Primatol, 48::10-21 2019.
- Hudson N, Celkova L, Hopkins A, Greene C, Storti F, Ozaki E, Fahey E, Theodoropoulou S, Kenna PF, Humphries MM, Curtis AM, Demmons E, Browne A, Liddie S, Lawrence MS, Grimm C, Cahill MT, Humphries P, Doyle SL, Campbell M. Dysregulated claudin-5 cycling in the inner retina causes retinal pigment epithelial cell atrophy. JCI Insight.4 2019.
VIRSCIO RESOURCES
Frequently Asked Questions
What normative databases exist for the St. Kitts African green monkey?
What normative databases exist for the St. Kitts African green monkey?
What are the shipping logistics and CITES requirements for samples from the St. Kitts African green monkey?
What are the shipping logistics and CITES requirements for samples from the St. Kitts African green monkey?
Although not endangered, as a nonhuman primate the St. Kitts African green monkey falls under the Convention on International Trade in Endangered Species (CITES) regulations. CITES permits are routinely obtained from the St. Kitts Ministry of Agriculture to enable shipment of green monkey specimens across the globe. Virscio maintains Fish and Wildlife Service (F&WS) and Center for Disease Control (CDC) permits for seamless importation to the U.S., and coordinates with relevant national authorities for shipments globally.
Cryopreserved samples are shipped in long hold time liquid nitrogen vapor shippers, ensuring sample integrity.